
ncert text book, class 12 physics, class 12 physics notes, physics class 12 notes, cbse 12 notes, nuclei physics , electromagnetic waves 12 notes, class 12 physics notes, electromagnetic waves class 12, electromagnetic waves class 12 notes, class 12 electromagnetic waves, note physics, physics notes, electromagnetic waves, class 12 cphysics chapter 8 notes, 12th standard physics notes, 12th std physics notes, class 12 physics notes chapter 8, electromagnetic waves chapter class 12 notes
| Thomson’s model of atom- Every atom consists of charged sphere in which electrons are embedded like seeds in water melon | Its drawbacks:-couldn’t explain large angle scattering & the origin of spectral series. |
Rutherford’s model of atom-
|
Limitations :-couldn’t explain the stability of the nucleus & the emission of line spectra of fixed frequencies.
|
| Distance of closest approach of the alpha particle in the α particle scattering experiment | |
| Impact parameter of the alpha particle | |
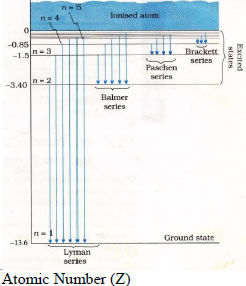
| Bohr’s model of atom | Limitations-applicable only for hydrogen like atoms & couldn’t explain the splitting of spectral lines. (not consider electro static force among the electrons) |
| Orbit radius of the electron around the nucleus | 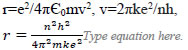 |
| Energy of the electron in the nth orbit of hydrogen atom | |
| Angular momentum of electron in any orbit is integral multiple of h/2ð | L = mvr = nh/2π, n=1,2,3,… |
| Wave number í | 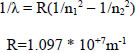 |
 |
No of protons in a nucleus |
| Mass Number (A) Number of neutrons | No. of nucleons(protons + neutrons) in a nucleus A-Z |
| Nuclear radius | R=R0 A1/3 |
| Nuclear density | Ñ= 3m/4ðR03 |
| Isotopes | Same Z & different A Ex, 1H2,1H3,1H1, & C12,C14,C16 |
| Isobars | Same A & different Z [ 18Ar40,20Co40] & (1H3, 2H3) |
| Isotones | Same no. of neutrons Mass of neutrons – 1H3, 2He4 |
| Mas defect Δ m | Total Mass of nucleons – mass of nucleus |
| Binding energy Eb | E= |
| Radioactive decay law | dN/dt=-ëN -dW/dt= R= Activity, unit Bq. |
| N =N0e-λt OR N=N0( ½)n , n = t/t1/2 |
|
| Half life | 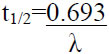 |
| Mean life | τ= 1/λ |
| 3 types of radiations | Alpha,beta,gamma |
| Nuclear fission | Splitting of a heavy nucleus into lighter elements.This process is made use of in Nuclear reactor & Atom bomb |
| Nuclear fusion Fusing | Fusing of lighter nuclei to form a heavy nucleus.This process takes place in Stars & Hydrogen bomb. Controlled Thermonuclear Fusion In a fusion reactor-high particle density is requiredhigh plasma temperature of 109Ka long confinement time is required |
Copyright @ ncerthelp.com A free educational website for CBSE, ICSE and UP board.