
haloalkanes and haloarenes class 12 ncert slolutions, haloarenes, electrochemistry notes, biology notes, biochemistry notes, biochemistry, biology notes for class 11, 2nd puc biology notes, photochemistry notes, xii class, chem notes, ncert test papers, online 12 exam, aliphatic and aromatic compound, ncert class 4, aliphatic and aromatic hydrocarbon, mcq test papers, aromatic test, an aromatic hydrocarbon , ncert solutions, chapter 10,chapter 10ncert solutions, haloalkanes and haloarenes ncert solutions, ncert solutions for class 12 chemistry, class 12 chemistry ncert solutions, ncert solutions for class 12, ncert class 12 chemistry, class 12 chemistry, class 12 chemistry solution, ncert solutions class 12, class 12 chemistry , ncert class 12, class 12 chemistry chapter 10,chapter 10
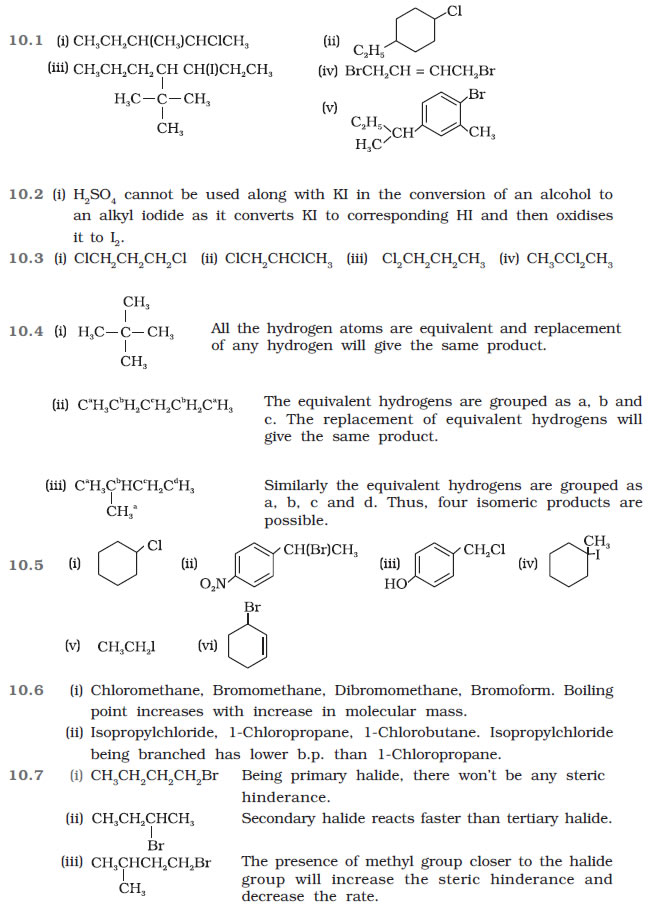
Question 10.1 Name the following halides according to IUPAC system and classify them as
alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
(i) (CH3)2CHCH(Cl)CH3
(ii) CH3CH2CH(CH3)CH(C2H5)Cl
(iii) CH3CH2C(CH3)2CH2I
(iv) (CH3)3CCH2CH(Br)C6H5
(v) CH3CH(CH3)CH(Br)CH3
(vi) CH3C(C2H5)2CH2Br
(vii) CH3C(Cl)(C2H5)CH2CH3
(viii) CH3CH=C(Cl)CH2CH(CH3)2
(ix) CH3CH=CHC(Br)(CH3)2
(x) p-ClC6H4CH2CH(CH3)2
(xi) m-ClCH2C6H4CH2C(CH3)3
(xii) o-Br-C6H4CH(CH3)CH2CH3
Question 10.2 Give the IUPAC names of the following compounds:
(i) CH3CH(Cl)CH(Br)CH3
(ii) CHF2CBrClF
(iii) ClCH2C≡CCH2Br
(iv) (CCl3)3CCl
(v) CH3C(p-ClC6H4)2CH(Br)CH3
(vi) (CH3)3CCH=ClC6H4I-p
Question 10.3 Write the structures of the following organic halogen compounds.
(i) 2-Chloro-3-methylpentane
(ii) p-Bromochlorobenzene
(iii) 1-Chloro-4-ethylcyclohexane
(iv) 2-(2-Chlorophenyl)-1-iodooctane
(v) Perfluorobenzene
(vi) 4-tert-Butyl-3-iodoheptane
(vii) 1-Bromo-4-sec-butyl-2-methylbenzene
(viii) 1,4-Dibromobut-2-ene
Question 10.4 Which one of the following has the highest dipole moment?
(i) CH2Cl2
(ii) CHCl3
(iii) CCl4
Question 10.5 A hydrocarbon C5H10 does not react with chlorine in dark but gives a single
monochloro compound C5H9Cl in bright sunlight. Identify the hydrocarbon.
Question 10.6 Write the isomers of the compound having formula C4H9Br.
Question 10.7 Write the equations for the preparation of 1-iodobutane from
(i) 1-butanol
(ii) 1-chlorobutane
(iii) but-1-ene.
Question 10.8 What are ambident nucleophiles? Explain with an example.
Question 10.9 Which compound in each of the following pairs will react faster in SN2 reaction
with –OH?
(i) CH3Br or CH3I
(ii) (CH3)3CCl or CH3Cl
Question 10.10 Predict all the alkenes that would be formed by dehydrohalogenation of the
following halides with sodium ethoxide in ethanol and identify the major alkene:
(i) 1-Bromo-1-methylcyclohexane
(ii) 2-Chloro-2-methylbutane
(iii) 2,2,3-Trimethyl-3-bromopentane.
Question 10.11 How will you bring about the following conversions?
(i) Ethanol to but-1-yne
(ii) Ethane to bromoethene
(iii) Propene to
1-nitropropane
(iv) Toluene to benzyl alcohol
(v) Propene to propyne
(vi) Ethanol to ethyl fluoride
(vii) Bromomethane to propanone
(viii) But-1-ene
to but-2-ene
(ix) 1-Chlorobutane to n-octane
(x) Benzene to biphenyl.
Question 10.12 Explain why
(i) the dipole moment of chlorobenzene is lower than that of cyclohexyl chloride?
(ii) alkyl halides, though polar, are immiscible with water?
(iii) Grignard reagents should be prepared under anhydrous conditions?
1
Question 10.13 Give the uses of freon 12, DDT, carbon tetrachloride and iodoform.
1
Question 10.14 Write the structure of the major organic product in each of the following reactions:
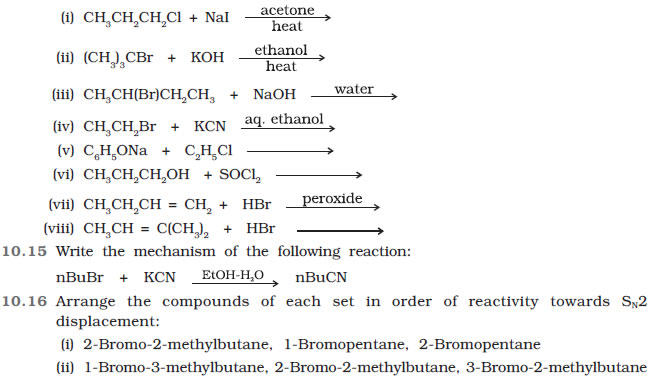
(iii) 1-Bromobutane, 1-Bromo-2,2-dimethylpropane, 1-Bromo-2-methylbutane,
1-Bromo-3-methylbutane.
Question 10.17 Out of C6H5CH2Cl and C6H5CHClC6H5, which is more easily hydrolysed by aqueous
KOH?
Question 10.18 p-Dichlorobenzene has higher m.p. and solubility than those of o- and m-isomers.
Discuss.
Question 10.19 How the following conversions can be carried out?
(i) Propene to propan-1-ol
(ii) Ethanol to but-1-yne
(iii) 1-Bromopropane to 2-bromopropane
(iv) Toluene to benzyl alcohol
(v) Benzene to 4-bromonitrobenzene
(vi) Benzyl alcohol to 2-phenylethanoic acid
(vii) Ethanol to propanenitrile
(viii) Aniline to chlorobenzene
(ix) 2-Chlorobutane to 3, 4-dimethylhexane
(x) 2-Methyl-1-propene to 2-chloro-2-methylpropane
(xi) Ethyl chloride to propanoic acid
(xii) But-1-ene to n-butyliodide
(xiii) 2-Chloropropane to 1-propanol
(xiv) Isopropyl alcohol to iodoform
(xv) Chlorobenzene to p-nitrophenol
(xvi) 2-Bromopropane to 1-bromopropane
(xvii) Chloroethane to butane
(xviii) Benzene to diphenyl
(xix) tert-Butyl bromide to isobutyl bromide
(xx) Aniline to phenylisocyanide
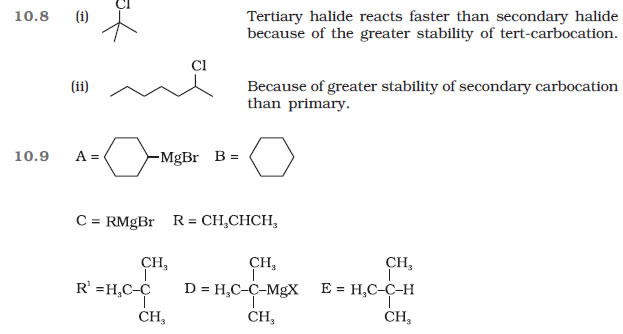
Question 10.20 The treatment of alkyl chlorides with aqueous KOH leads to the formation of
alcohols but in the presence of alcoholic KOH, alkenes are major products. Explain.
Question 10.21 Primary alkyl halide C4H9Br
(a) reacted with alcoholic KOH to give compound
(b).
Compound
(b) is reacted with HBr to give
(c) which is an isomer of (a). When
(a) is reacted with sodium metal it gives compound
(d), C8H18 which is different
from the compound formed when n-butyl bromide is reacted with sodium.
Give the structural formula of (a) and write the equations for all the reactions.
Question 10.22 What happens when
(i) n-butyl chloride is treated with alcoholic KOH
(ii) bromobenzene is treated with Mg in the presence of dry ether,
(iii) chlorobenzene is subjected to hydrolysis,
(iv) ethyl chloride is treated with aqueous KOH,
(v) methyl bromide is treated with sodium in the presence of dry ether,
(vi) methyl chloride is treated with KCN?
Please Wait pdf file is loading (कृपया इंतजार करें pdf file लोड हो रही है)...
Loading speed will depend up on your download speed. Pdf file के लोड होने में लगा समय आपकी डाउनलोड स्पीड पर निर्भर करेगा
Copyright @ ncerthelp.com A free educational website for CBSE, ICSE and UP board.