
saturated hydrocarbons, saturated and unsaturated hydrocarbons, saturated hydrocarbon, saturated and unsaturated hydrocarbon, saturated aliphatic hydrocarbon, saturated hydrocarbon and unsaturated hydrocarbon, define saturated hydrocarbon, hydrocarbon saturation , hydrocarbons 11 notes, class 11 chemistry notes, hydrocarbons class 11, hydrocarbons class 11 notes, class 11 hydrocarbons, note chemistry, chemistry notes, hydrocarbons, class 11 cchemistry chapter 13 notes, 11th standard chemistry notes, 11th std chemistry notes, class 11 chemistry notes chapter 13, hydrocarbons chapter class 11 notes
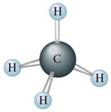
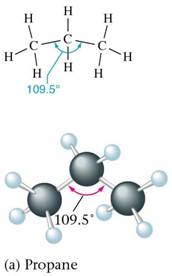
➣ Hydrocarbons are composed of Carbon and hydrogen.
➣ The important fuels like Petrol, kerosene, coal gas, CNG, LPG etc. are all hydrocarbons or their mixture.
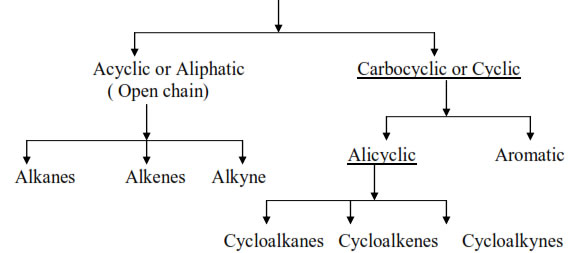
Sources:
Petroleum and natural gas are the major sources of aliphatic hydrocarbon while coal is an important source of aromatic hydrocarbons. The oil trapped inside the rocks is known as
petroleum. PETRA – ROCK, OLEUM – OIL. The oil in the petroleum field is covered with
a gaseous mixture known as natural gas. The main constituents of the natural gas are methane, ethane, propane and butane.
Cycloalkanes Cycloalkenes Cycloalkynes C–C bond
➣ Chemically unreactive
➣ Show chain, position and optical isomerism.
➣ Heptane has 9 isomer, Octane 18 and Decane 75. Nomenclature:
➣ |
Follow mainly free radical mechanism |
|
||
➣
➣ |
Useful in preparing Stepping up reaction |
an |
alkane |
containing |
![]()
.jpg)
4. From carboxylic acids :- Decarboxylation

(1) Nature:- Non-Polar due to covalent nature of C—C bond and C—H bond. C—
C bond enrgy = 83 kj/mole and C—H bond energy = 99 kj/mole. C1—C4 = gases, C5—C17 = colourless odourless liq uid and > C17 = Solid.
(2) Solubility :- Like dissolve like
Viz, Polar compounds dissolve in polar solvent and Non-Polar compound dissolve in non polar solvent.
(3) Boiling point :- Low boiling point due to non polar in nature The molecules are held together only by weak Van der Waalls’ forces.
Since we known that the magnitude of Van der Waalls’ forces is directly proportional to the molecular size. Therefore, the boiling point increases with increase the molecular size i.e. with increase in number of carbon atoms.
Noted :- the boiling points of the branched chain Alkanes are less than the straight chain isomers.
This is due to the fact that branching of the chain makes the molecule more
compact and thereby decreases the surface aria and consequently, the magnitudes of
Van der Waalls’ forces also decrease.
![]()
n-penatne (boling point =309k
regular variation with ven number of carbon atoms having higher melting point a 2O2 CO2 2H2O lkanes having immediately next lower and immediately next higher odd number of carbon atoms.
Noted :- Iodination is a reversible reaction. So it is carried out by heating alkane in the presence of some oxidizing agent like iodic acid (HIO3) or nitric acid (HNO3) or mercuric oxide (HgO) which oxidizes HI formed during the reaction.
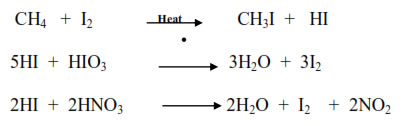
Noted :- Fluorination of alkane takes place explosively resulting even in the rupture of C—C bond in higher alkanes.
(i) The reactivity of Halogens:- F2 > Cl2 > Br2 > I2.
(ii) The rate of replacement of Hydrogens of alkanes is :- 3° > 2° > 1°
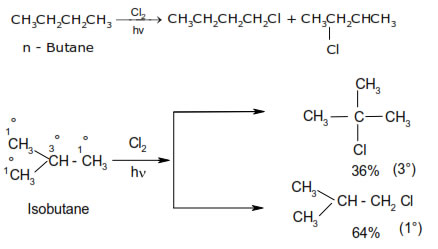
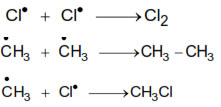
Mechanism :- halogenations reaction take place by free radical mechanism. The reaction proceeds in the following steps :
Initiation
(i) Chain initiation step:-
![]()
(ii) Chain Propagation step :-
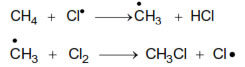
(iii) Chain Termination step :-
• At high temp C—C bond is also broken so that mixture of nitroalkanes is obtained.
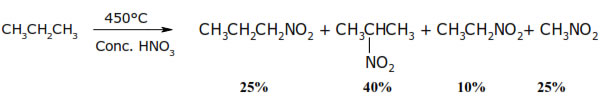
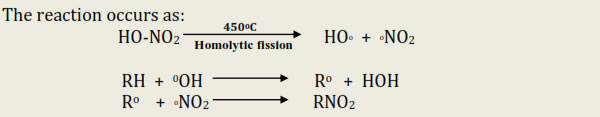
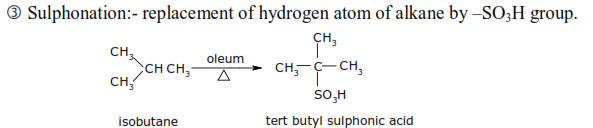
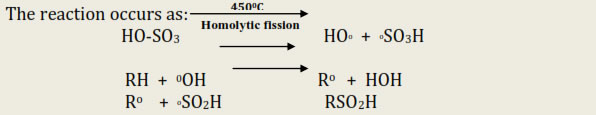
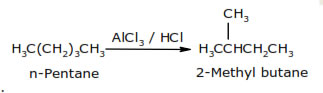
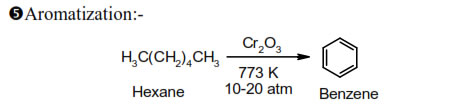
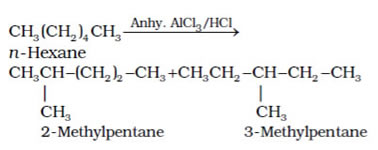
This method is also called dehydrogenation or hydroforming
Similarly, heptane gives toluene, n-Octane give o-xylene and 2, methyl heptane give m-xylene.
![]() Thermal decomposition or Pyrolysis or cracking or Fragmentation: - when higher
alkanes are heated at high temp (about 700-800k) in the presence of alumina or silica catalysts, the alkanes break down to lower alkanes and alkenes.
Thermal decomposition or Pyrolysis or cracking or Fragmentation: - when higher
alkanes are heated at high temp (about 700-800k) in the presence of alumina or silica catalysts, the alkanes break down to lower alkanes and alkenes.
CH3-CH2-CH3 ![]() CH3-CH-CH2 + CH3-CH3 + C2H4 + CH4
CH3-CH-CH2 + CH3-CH3 + C2H4 + CH4
![]() Action of steam :- catalyst: nickel, alumina Al2O3
1000 oC CH4 + H2O(Steam)
Action of steam :- catalyst: nickel, alumina Al2O3
1000 oC CH4 + H2O(Steam) ![]() CO + 3H2
CO + 3H2
This reaction is used for the industrial preparation of hydrogen from natural gas.
8. Isomerisation :-
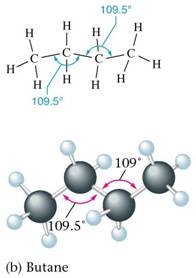
The different molecular arrangements arising as a result of rotation around carbon carbon single bonds are called conformational isomers or rotational isomers and the phenomenon is called conformational isomerism.
Numerous possible arrangements of ethane are possible. Two extreme conformations are known. These are eclipsed conformation and staggered conformation.
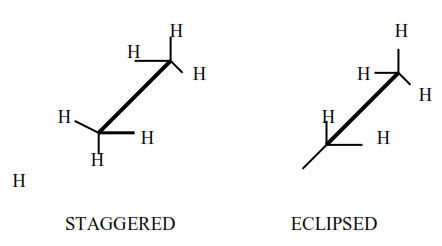
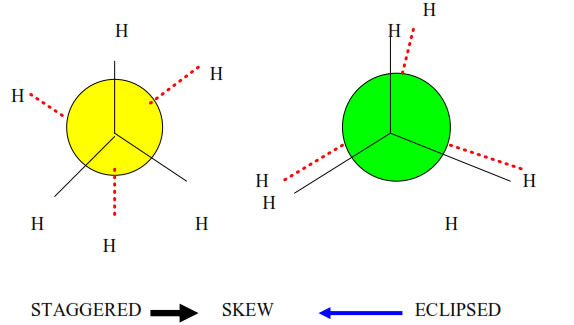
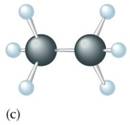
• Unsaturated hydrocarbon which have double bond.
• General molecular formula CnH2n
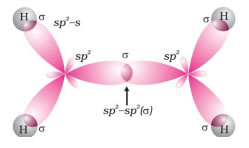
• C–C bond hybridization 1.34 A0
• sp2 hybridization
• When we treated Alkene with chlorine, oily products are obtained. So Alkenes are also known as Olefins. (Greek olefiant meaning oil forming).
• Show chain, positional and geometrical isomerism
➣ Structure of double bond:
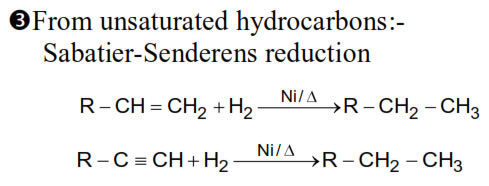
1. From Alkynes :- Alkynes on partial reduction with Partially deactivated palladised charcoal known as Lindlar’s catalyst give alkynes.
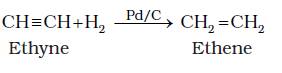
2. From Haloalkanes : - dehydrohalogenation
(E2 or 1,2-elimination or Bita-elimination)
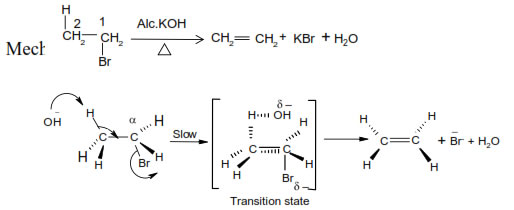
➣ predominant formation of a substituted alkene is formed according to Saytzeff’s rule
3. From Dihaloalkane s: - dehalogenation
4. From Alcohols: - Dehydration

Addition Reaction:- Alkene show electrophilic addition reaction
.
1. Addition of Hydrogen:-
3. Addition of hydrogen halides :- Addition reaction of HBr to symmetrical alkenes
![]() Addition reaction of HBr to unsymmetrical alkenes takes place according to
Markovnikov Rule
Addition reaction of HBr to unsymmetrical alkenes takes place according to
Markovnikov Rule
negative part of the addendum (adding molecule) gets attached to that carbon atom which possesses lesser number of hydrogen atoms. e g
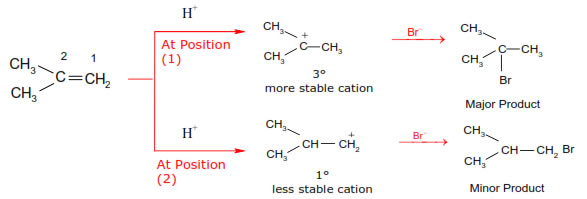
In 1933 Kharasch and Mayo observed that when HBr is added to an unsymmetrical double bond in the presence of organic peroxide, the reaction take places opposite to the Markovnikov rule.
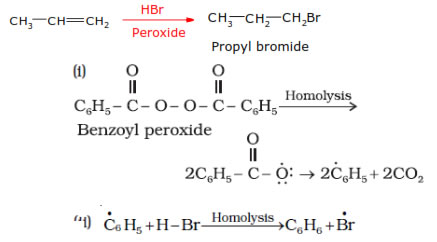
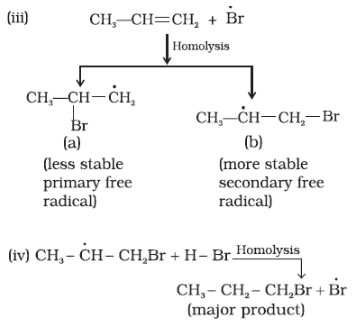
Noted:- peroxide effect is applicable only to HBr and not to HF, HCl and HI. Addition of HF, HCl and HI takes place according to Markovnikov’s rule even in the presence of peroxide.
4. Addition of water (Hydration) :- Acid catalyzed addition of water
.jpg)
![]() Oxidation:-
Oxidation:-
1.Combustion :- CO2
+ H2
O
2.Hydroboration–oxidation :- Alkanes react with diborane to form trialkyl boranes
which on oxidation with alkaline H2O2 give alcohols.
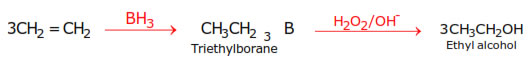
3.Oxymercuration–demercuration:-
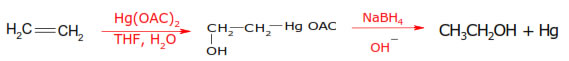
4.Oxidation with potassium permanganate:-
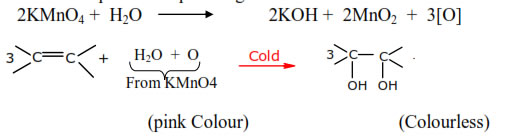
➣ This reaction is also called Hydroxylation
➣ Cis product I.e. cis-diol is obtained.
Noted :- The alkaline potassium permanganate solution is known as Baeyer’s reagent.
It has bright pink colour. It oxidizes alkenes to glycols which is colourless. This
reaction is used as a test for the presence of double bond in a molecule. This is also
known as Baeyer test.
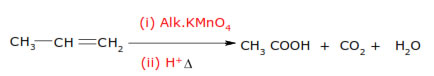
☺Oxidation with Ozone :- Ozonolysis – give carbonyls compounds
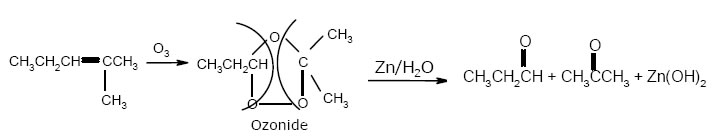
Noted:- Bromine water test and Baeyer’s test are used to detect the presence of double bond while ozonolysis is used to detect the position of double bond.
• Unsaturated hydrocarbon which have triple bond.
• General molecular formula CnH2n–2
• sp hybridization
• Shows chain, positional and functional isomerism
• Preparation:-
From vicinal dihalides : - dehalogenation
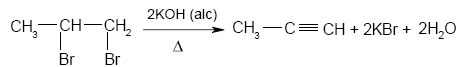
By the action of water on calcium carbide:-
![]()
➣ Chemical Properties:-
![]() Addition Reaction:- Alkyne show electrophilic addition reaction
Addition Reaction:- Alkyne show electrophilic addition reaction
1.
Addition of Hydrogen:- Hydrogenation.
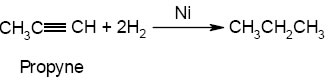
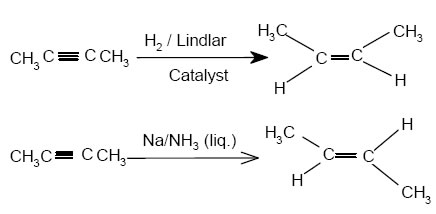
Noted:- It may be noted that the hydrogenation can be controlled at the alkene stage only. This is possible by using a Lindlar’s catalysts or sodium in liquid NH3 at 200k temp..
Noted:- It may be again noted that the catalytic reduction of alkynes in the presence of Lindlar’s catalyst gives cis-alkenes while in the presence of sodium in liquid NH3 (Birch reduction) gives trans-alkenes.
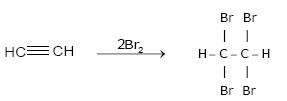
2.Addition of Halogens:-
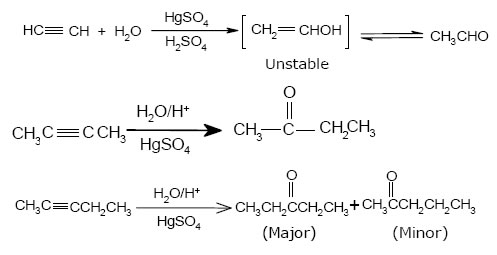
3.Addition of hydrogen halides:-
![]()
4.Addition of water (Hydration):- Acid catalyzed addition of water
5. Polymerisation-
a. Linear polymerisation: of ethyne gives polyacetylene or polyethyne which is a high molecular weight polyene containing repeating units of (CH = CH – CH = CH ) and can be represented as —(CH = CH – CH = CH)n —
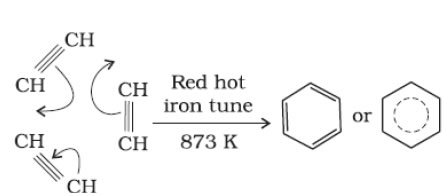
b. Cyclic polymerization- results in the formation of aromatic compound.
Acidity of Alkynes- Terminal alkynes are acidic in nature
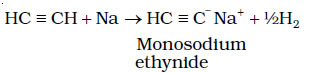
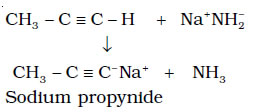
Alkanes, alkenes and alkynes follow the following trend in their acidic behaviour :
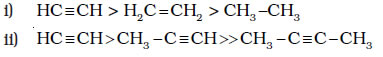
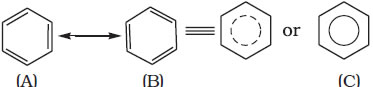
Aromatic compounds containing benzene ring are known as benzenoids and those not containing a benzene ring are known as non-benzenoids. Structure of Benzene- Kekulé structure
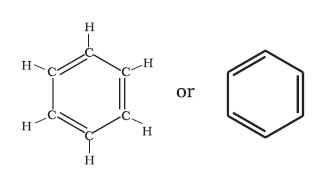
Resonance and stability of benzene-Benzene is a hybrid of various resonating structures.
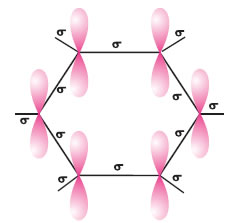
The orbital overlapping picture benzene- All the six carbon atoms in benzene are sp2 hybridized and these hybrid orbitals form sigma bonds.
The unhybridised p orbital of carbon atoms are close enough to form a π bond by lateral overlap.
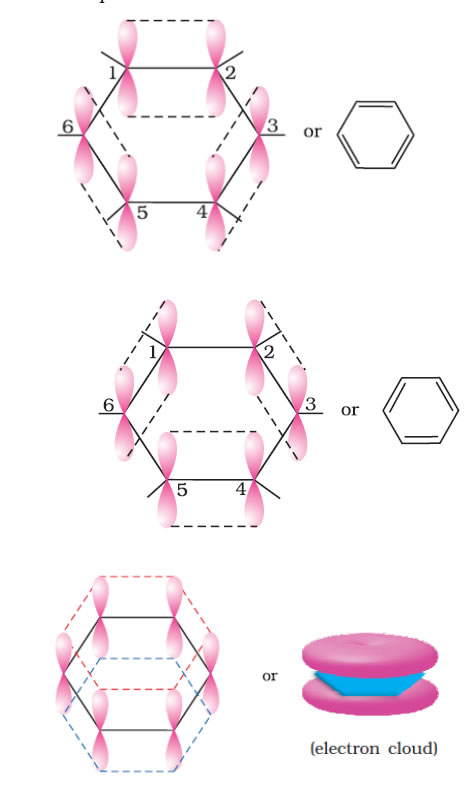
The six π electrons are thus delocalised and can move freely about the six carbon nuclei. The delocalised π electron cloud is attracted more strongly by the nuclei of the carbon atoms than the electron cloud localized between two carbon atoms. Therefore, presence of delocalised π electrons in benzene makes it more stable .
The compounds that follow the following features are to be considered aromatic.
(i) Planarity
(ii) Complete delocalisation of the π electrons in the ring
(iii) Presence of (4n + 2) π electrons in the ring where n is an integer (n = 0, 1,
2, . . .). This is often referred to as Hückel Rule
(i) Cyclic polymerisation of ethyne
(ii) Decarboxylation of aromatic acids
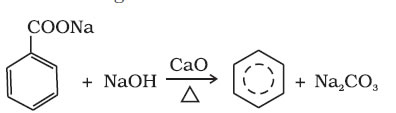
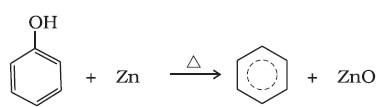
(iii) Reduction of phenol: Phenol is reduced to benzene by passing its vapours over heated zinc dust
Physical properties:
1. Aromatic hydrocarbons are non- polar molecules and are usually colourless liquids or solids with a characteristic aroma.
2. Aromatic hydrocarbons are immiscible with water but are readily miscible with organic solvents.
3. They burn with sooty flame.
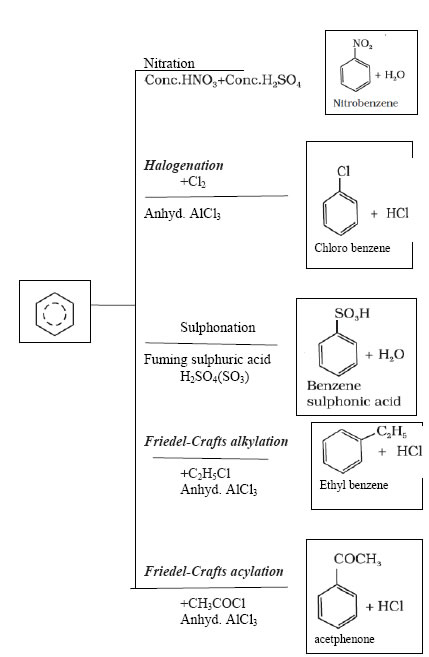
Arenes are characterised by electrophilic substitution reactions proceed via the following three steps:
(a) Generation of the eletrophile
(b) Formation of carbocation intermediate
(c) Removal of proton from the carbocation intermediate

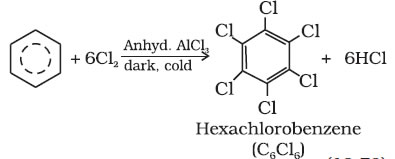
benzene ontreatment with excess of chlorine in the presence of anhydrous AlCl3 in dark yields hexachlorobenzene (C6Cl6)
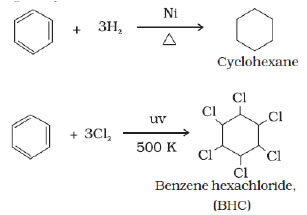
Addition reactions of benzene-
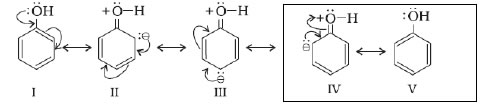
Directive influence of a functional group in monosubstituted benzene:-
1. Ortho and para directing groups and activating :- –OH, –NH2, –NHR, –NHCOCH3, –OCH3, –CH3, –C2H5, etc.
2. Meta directing group and deactivating:–NO2, –CN, –CHO, –COR, –COOH, –COOR, –SO3H, etc.
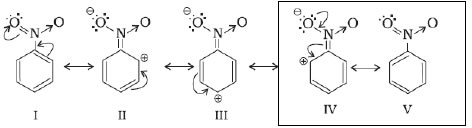
3. Ortho and para directing groups and deactivating :- Halogens because of their strong – I effect, overall electron density on benzene ring decreases. However, due to resonance the electron ensity on o– and p– positions is greater than that at the m-position. Hence, they are also o– and p– irecting groups
CARCINOGENICITY AND TOXICITY-Benzene and polynuclear hydrocarbons containing more than two benzene rings fused together are toxic and said to possess cancer producing (carcinogenic) property.
Copyright @ ncerthelp.com A free educational website for CBSE, ICSE and UP board.