
class 9 science notes, chapter 4 structure of the atom , structure of the atom 9 notes, class 9 science notes, structure of the atom class 9, structure of the atom class 9 notes, class 9 structure of the atom, note science, science notes, structure of the atom, class 9 cscience chapter 4 notes, 9th standard science notes, 9th std science notes, class 9 science notes chapter 4, structure of the atom chapter class 9 notes

• More than 2400 years ago, he named the smallest piece of matter “ATOMOS ,” meaning “not to be cut.” To Democritus,
• Atoms were small, hard particles that were all made of the same material but were different shapes and sizes.
• Atoms were infinite in number, always moving and capable of joining together1. Dalton’s Atomic theory:

• First recorded evidence that atoms existed.
• Using his theory, Dalton rationalized the various laws of chemical combination
Dalton’s theory was based on the premise that the atoms of different elements could be distinguished by differences in their weights.
• Limitations
o The indivisibility of an atom was proved wrong , for, an atom can be further subdivided into protons, neutrons and electrons.
o The atoms of same element are similar in all respects , but isotopes of same element have different mass.
o Dalton’s theory was based on the premise that the atoms of different elements could be distinguished by differences in their weights.

Discovered electrons in 1897.
• Showed us that the atom can be split into even smaller parts.
His discovery was the first step towards a detailed model of the atom .
• An atom is a uniform sphere of positive charges (due to presence of protons) as well as negative charges (due to presence of electrons).
• An electron is a negatively charged component of an atom which exists outside the nucleus. Each electron carries one unit of negative charge and has a very small mass as compared with that of a neutron or proton.
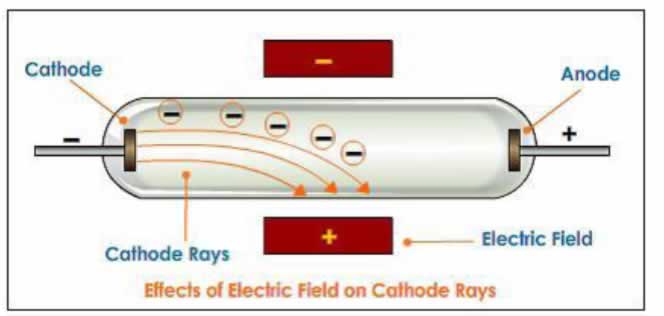
JJ Thomson used cathode ray tubes to demonstrate that the cathode ray responds to both magnetic and electric fields.
Since the ray was attracted to a positive electric plate placed over the cathode ray tube (beam deflected toward the positive plate) he determined that the ray must be composed of negatively charged particles. He called these negative particles "electrons."
Limitation: Model failed to explain how protons and electrons were arranged in atom so close to each other.
Eugene Goldstein:
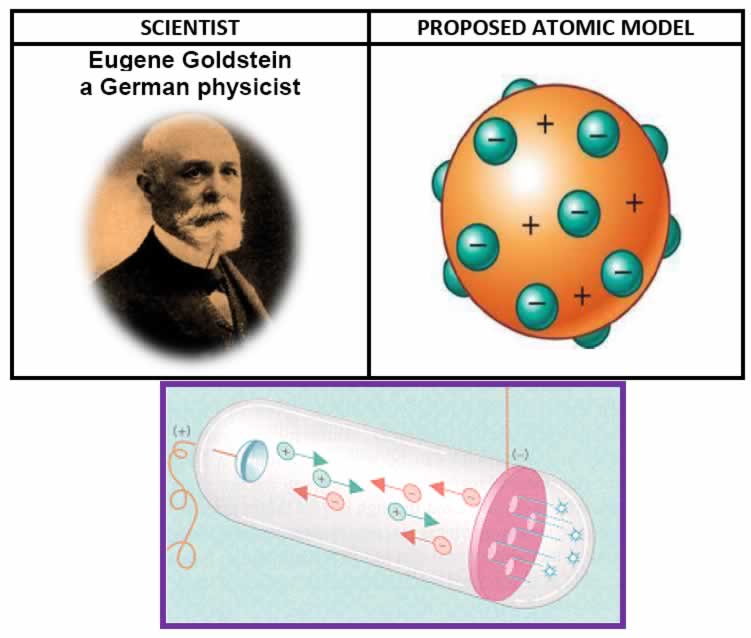
"canal rays" which had electrical and magnetic properties opposite of an electron
Protons:
The canal rays have positively charged sub-atomic, particles known as protons (p).
Q.1 What was the model of an atom proposed by Thomson?
Q.2 What is the nature of charge on electrons?
Q.3 What are canal rays ?
Q.4 State the nature of the constituents of canal rays.
3. Rutherford’s Scattering Experiments:

Experiment: Rutherford took a thin gold foil and made alpha particles , [ He2+ ] positively charged Helium fall on it.
S.No |
OBSERVATION |
INFERENCE |
1. |
Most of the a-particles passed through the gold foil without getting deflected. Very few particles were deflected. | Most of the space inside the atom is empty. |
2. |
Very few particles were deflected. | Positive charge of the atom occupies very little space. |
3. |
A very few alpha particles, 1 in 100000 completely rebound on hitting the gold foil. | Nucleus of an atom is very small as compared to the total size. |

Atomic radius ~ 100 pm = 1 x 10-10 m
Nuclear radius ~ 5 x 10-3 pm = 5 x 10-15 m
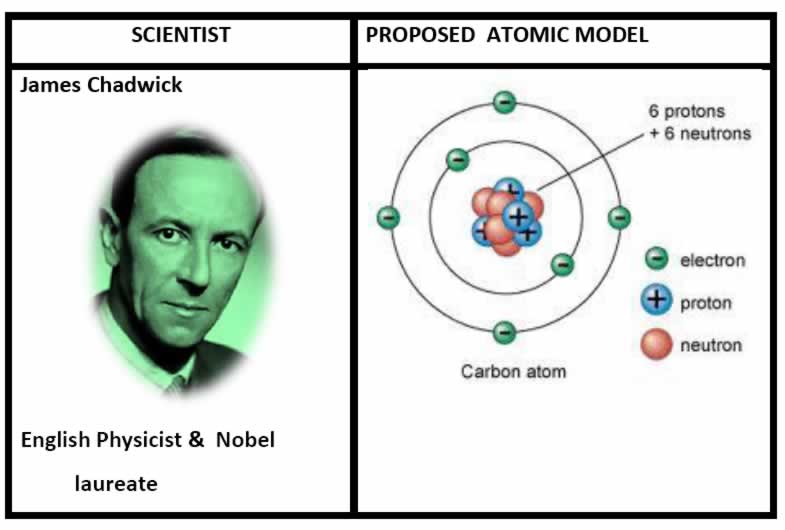
• In 1932, James Chadwick proved that the atomic nucleus contained a neutral particle which had been proposed more than a decade earlier by Ernest Rutherford officially discovered the neutron in 1932, Chadwick received the Nobel Prize in 1935.
A neutron is a subatomic particle contained in the atomic nucleus. It has no net electric charge, unlike the proton’s positive electric charge.
Q.1 Who discovered the nucleus of the atom?Q.2 What is the charge on alpha particles ?
Q.3 Which observation of Rutherford’s scattering experiment established the presence
large empty space in atom?
Q.4 What is the nature of charge on nucleus of atom?
Q.5 Who discovered neutron ?
4. Sub Atomic Particles:
| Name | Symbol | Location in the atom | Charge | Relative Mass | Actual Mass (g) |
| Electron | E | Around the nucleus | 1- | 1/1840 | 9.11 x 10 -28 |
| Proton | P | In the nucleus | 1+ | 1 | 1.67 x 10 -24 |
| Neutron | n | In the nucleus | 0 | 1 | 1.67 x 10 -24 |
Protons & Neutrons collectively are known as NUCLEONS.
Q.1 Why is the relative mass of an electron is taken as negligible ?
Q.2 Give the actual masses of electron & proton in kg?
Q.3 What are nucleons?
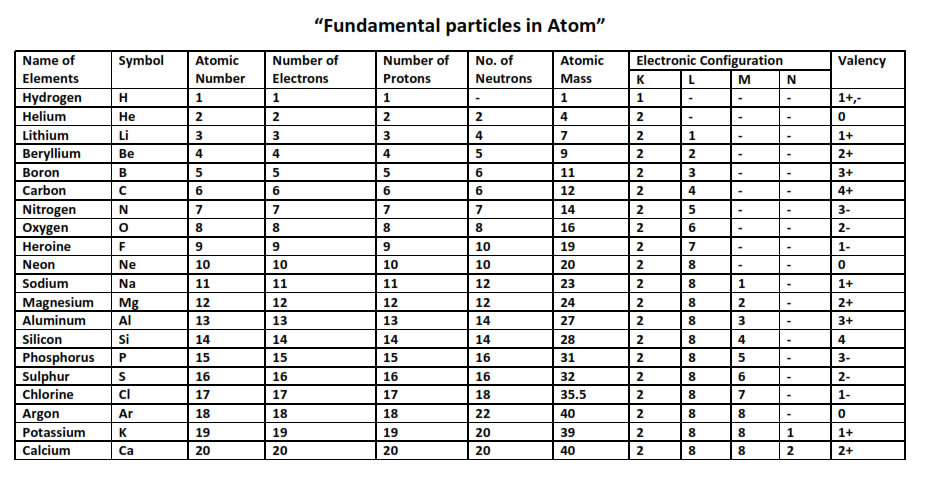
5.Atomic Number & Mass Number:
“Atomic number of an element is defined as the number of unit positive charges on the nucleus (nuclear charge) of the atom of that element or as the number of protons present in the nucleus.”
Atomic number, Z = Number of unit positive charge on the nucleus = Total number of unit positive charges carried by all protons present in thenucleus. = Number of protons in the nucleus (p) = Number of electrons revolving in the orbits (e)
Eg :- Hydrogen – Atomic number= 1 (1 proton)
Helium - Atomic number = 2 (2 protons)
Mass number[ A] : It is defined as the sum of the number of protons & neutrons present in the nucleus of an atom.
Mass Number = Mass of protons + Mass of neutrons
Eg :- Carbon – Mass number = 12 (6 protons + 6 neutrons) Mass = 12u Aluminium – Mass number
= 27 (13 protons + 14 neutrons) Mass = 27
Q.1 The mass number of an element is 18. It contains 7 electrons .What is the number of
protons and neutrons in it ?
Q.2 An atom contains 3 protons , 3 electrons and 4 neutrons .
i) What is its atomic number ? ii) What is its mass number?
Q.3 An element is represented by
. Give the number of electrons and neutrons in it.
6. Niel Bohr Atomic Model:
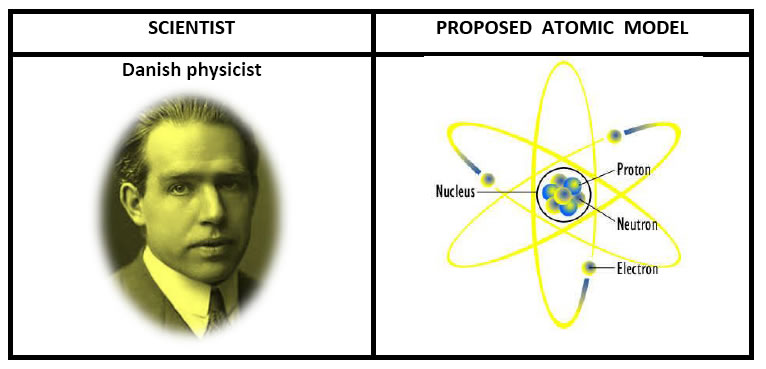
Main Postulates of the Bohr Model [refer NCERT Text Book article 4.3 ,page number-49]
Q.1 What happens when an electron jumps from lower to higher energy level?
Q.2 Which energy shell is nearest to the nucleus of an atom?
Q.3 Which energy shell has higher energy L or N ?
7. Electronic configuration & Valency:
Bohr and Bury Scheme - Important Rules
S.No |
Electron Shell |
2n2
where n = shell number |
Maximum Capacity |
1 |
K Shell |
2 x (1) 2 |
2 electrons |
2 |
L Shell |
2 x (2) 2 |
8 electrons |
3 |
M shell |
2 x (3) 2 |
18 electrons |
4 |
N shell |
2 x (4) 2 |
32 electrons |
The outermost shell of an atom cannot accommodate more than 8 electrons, even if it has a capacity to accommodate more electrons. This is a very important rule and is also called the OCTET RULE. The presence45 of 8 electrons in the outermost shell makes the atom very stable.
Q.1 An atoms has atomic number 13. What would be its configuration.
Q.2 What is octet rule?
Q.3 How many electrons M shell can accommodate?
Q.4 If an atom has complete K and L shell, what would be its atomic number?
8. Isotopes & Isobars:
| ISOTOPES | ISOBARS | chemically same , physically different | Chemically different , physically same | Number of electrons is same | Number of electrons is different . | Cannot be separated by chemical means | Can be separated by chemical means |
QUESTION BANK [ *HOTS ]
1 Mark Questions:
1. Write the names of three elementary particles which constitute an atom.
2. Name the scientist & his experiment to prove that nucleus of an atom is positively
charged.
3. Which is heavier , neutron or proton ?
4. *How many times a proton is heavier than an electron?
5. What was the model of an atom proposed by Thomson ?
6. How many electrons at the maximum can be present in the first shell ?
7. What type of charge is present on the nucleus of an atom?
8. Give the number of protons in 35Cl17
9. *What are iso bars ?
10. Name the particles which determine the mass of an atom.
2 Marks Questions:
1. Define the following terms: a) Atomic number b) Mass number
2. Write the charges on sub atomic particles.
47
3. Identify the isotopes out of A , B , C & D ? 33A17 , 40B20 , 37C
17 , 38D19
4.* Give one Achievement and one limitation of J.J Thomson’s model of atom?
5. What are valence electrons? Give example.
6. *Which kind of elements have tendency to lose electron ? Give example.
7. How many electrons are present in the valence shell of nitrogen & argon?
8. State the maximum capacity of various shells to accommodate electrons.
9. Give the symbol , relative charge & mass of the three sub atomic particles.
32 S16 state :
i) Atomic number of sulphur, ii) Mass number of sulphur
iii) Electronic configuration of sulphur.
3. Marks Questions:
1. Why do Helium has Zero valency?
2. An atom contains 3 protons , 3 electrons and 4 neutrons .What is its atomic number , mass
number & valency?
3. *How are the isotopes of hydrogen represented ?
4. Write the complete symbol for the atom with the given atomic number [Z] & mass
number[A].
| i)Z=17,A=15; | ii)Z=4,A=9; | iii)Z=92;A=233 |
5 Marks Questions:
1.* a) Give the observations as well as inferences of Rutherford’s Scattering experiment for determining the structure of an atom.
b) On the basis of above experiment write the main features of atomic model.
2. Write the main postulates of Bohr’s Model of Atom.
• Rutherford established the existence of nucleus.
• Difference between Atomic number and Mass number
• Electronic configuration & its relation with Valency.
• Difference between Isotope and Isobar.
MULTIPLE CHOICE QUESTIONS ON CHEMISTRY PRACTICALS
1. To prepare
a) a true solution of common salt, sugar and alum
b) a suspension of soil, chalk powder and fine sand in water
c) a colloidal of starch in water and egg albumin in water and distinguish between these on
| the basis of | i) transparency | ii) filtration criterion | iii) stability |
1. Name the solute in common salt solution :
| a) H2O | NH4Cl |
| c) NaOH | d) NaCl |
2 Pick out a colloid from the following :
3. Which of the following is property of true solution ?
| a)Homogeneous | b) Heterogeneous |
| c) Translucent | d) None of these |
4.The process of separation of insoluble solids from a liquid is called :
| a) Filtration | b) Decantation |
| c) Sedimentation | d) Evaporation |
5. Which of the following mixtures is stable?
| a) Milk in water | b) Sugar in water |
| c) Sand in water | d) Wheat flour in cold water |
6. Egg albumin in water forms :
| a) White | b) Colourless |
| c) Green | d) Yellow |
7. Which of the following represents a correct set of observations for a mixture of common salt and water
| Transparency | Stability | Filtration |
| a) Transparent | Unstable | No residue |
| b) Transparent | Stable | No residue |
| c) Translucent | stable | No residue |
| d) Opaque | Unstable | Residue |
2. To prepare a) a mixture b) a compound
using iron filings and sulphur powder and distinguish between these on the basis of:
i. appearance i.e., homogeneity and heterogeneity
ii. behaviour towards a magnet
iii. behaviour towards carbon disulphide as a solvent.
iv. effect of heat.
1.The colour of sulphur is :
| a) White | b) Colourless |
| c) Green | d) Yellow |
| a) It has lost properties of its components. | b) It is not made up of iron. |
| c) It is a mixture | d) It is black in colour. |
| a) Visible | b) Not visible |
| c) Visible under microscope | d) None of these |
| a) High temperature | b) Low temperature |
| c) Moderate temperature | d) Below 0oC |
| a) Black to yellow | b) Yellow to black |
| c) Greyish yellow to black | d) Black to brown |
| a) Iron | b) Sulphur |
| c) Iron sulphide | d) Carbon |
| a) Blue | b)Colourless |
| c) Brown | d) Yellow |
2. What happens when Zn granules react with dilute sulphuric acid :
| ) Bubbles due to colourless , odourless gas are formed and colourless solution is obtained . | b) No reaction takes place. | c) Pungent smelling gas comes out. | d) No gas evolved. |
3. Fe + CuSO4 → FeSO4 + Cu is an example of :
| a) Displacement reaction | b) Decomposition reaction |
| c) Redox reaction | d) Double decomposition |
| a) The solution becomes pale green and reddish brown copper metal gets deposited. | b) The solution becomes colourless |
| c) There is no reaction | d) Copper displaces iron |
| a) White ppt | b) Colourless |
| c) Blue solution | d) Blue ppt. |
| a) It remains red | b) It turns blue |
| c) It becomes white | d) It becomes black |
| a) Acidic | b) Basic |
| c) Neutral | d) All of these |
4. To separate the components of a mixture of sand , common salt and ammonium chloride (or camphor) by sublimation.
1. Sublimation is the process in which :
a) Liquid changes into gaseous state
b) Solid changes into liquid state
c) Solid directly changes into gaseous form
d) Solid first converts into liquid , then into vapour form.
53
2. Which of the following substances sublimes on heating :
a) Iodine
b) Camphor
c) Naphthalene
d) All of these
3. In a mixture of sand, common salt and ammonium chloride , the substance that sublimes is:
a) Ammonium chloride
b) Sand
c) Common salt
d) All of these
4. Recovery of salt from salt solution in water can be done by :
a) Evaporation
b) Distillation
c) Filtration
d) None of these
5. What do you observe in the inner side of the funnel during sublimation of NH4Cl
a) Particles of white solid
b) Yellow fumes
c) Vapours of sodium chloride
d) No fumes are deposited
6. Which of the following mixtures cannot be separated by sublimation :
a) Ammonium chloride & sodium chloride
b) Ammonium chloride & sand
c) Ammonium chloride & iodine
d) Ammonium chloride & copper sulphate
7. A mixture of common salt and ammonium chloride is heated in a china dish covered with
an inverted funnel with stem closed with a cotton plug. After the experiment the china
dish will contain :
a) Common salt
b) Ammonium chloride
c) Both (a) and (b)
d) None of these.
5. To determine the melting point of ice and the boiling point of water.
1. Which of the following will help in determining the melting point of ice accurately ?
a) Ice made from tap water
b) Ice made from distilled water
c) Ice made mixed with salt
d) None of these
54
2. In determination of melting point of ice , the ice is contaminated with some non-volatile
impurity, like common salt, melting point of ice will :
a) Increase
b) Decrease
c) May increase or decrease
d) Does not change
3. What is the melting point of ice?
a) 0oC
b) 100oC
c) 273 K
d) Both (a) and (c)
4. Which vessel is used to determine the melting point of ice ?
a) Beaker
b) R B Flask
c) Conical Flask
d) Measuring Flask
5. At what temperature ice and water both exist together under normal atmospheric pressure ?
a) Below 273.16 K
b) Above 273.16 K
c) At 273.16 K
d) None of these
6. In determining the boiling point of water , correct reading is obtained when :
a) Temperature start rising
b) Water starts boiling
c) Whole of water evaporates
d) Temperature becomes constant
7.Water evaporates faster :
a) In still air
b) In humid air
c) In dry air
d) In windy & dry air
6. To verify the law of conservation of mass in a chemical reaction.
1. What does the law of conservation of mass state?
a) It states that mass is neither created nor destroyed.
b) It states that mass can be created or destroyed.
c) It states that mass cannot be created but can be destroyed.
d) It states that mass can be created but cannot be destroyed.
55
2. If you melt 100 g of ice will you get the same mass of water ?
a) Yes
b) No
c) May be
d) Sometimes
3. State the chemical reaction between Barium Chloride (aqueous) & Sodium Sulphate (aqueous) .
a) BaCl2 (aq) + Na2SO4 (aq) → BaSO4 (white ppt) + 2NaCl (aq)
b) BaCl2 (aq) + Na2SO4 (aq) → BaSO3 (red ppt) + 2NaCl (aq)
c) Both (a) & (b)
d) None of these
4. Which of the following reaction does not conform to law of conservation of mass ?
a) Burning of candle
b) Melting of ice
c) Fusion reaction occurring in sun
d) Combustion of fuel
5. In the reaction 2NaN2 .→ 2Na +3N2 , if 850 g of NaN2 is decomposed to give 265.20 g of
Na , how much N2 is produced ?
a) 584.80 g
b) 265.20 g
c) 850 g
d) 484.20 g
6. In chemical reactions how does law of conservation of mass contribute in writing
chemical equations ?
a) It does not help.
b) Equations can be balanced by writing their correct co –
efficient
c) Both (a) and (b)
d) None of these
7. In accordance with the law of conservation of mass give the co-efficient of O2 in the
equation:
C5H12+ O2 → 5CO2 + 6H2O
a) 4
b) 6
c) 8
d) 2
Copyright @ ncerthelp.com A free educational website for CBSE, ICSE and UP board.