
The new and updated curriculum is based on disciplinary approach with rigour and depth taking care that the syllabus is not heavy and at the same time it is comparable to the international level. The knowledge related to the subject of Chemistry has undergone tremendous changes during the past one decade. Many new areas like synthetic materials, bio -molecules, natural resources, industrial chemistry are coming in a big way and deserve to be an integral part of chemistry syllabus at senior secondary stage. At international level, new formulations and nomenclature of elements and compounds, symbols and units of physical quantities floated by scientific bodies like IUPAC and CGPM are of immense importance and need to be incorporated in the updated syllabus.
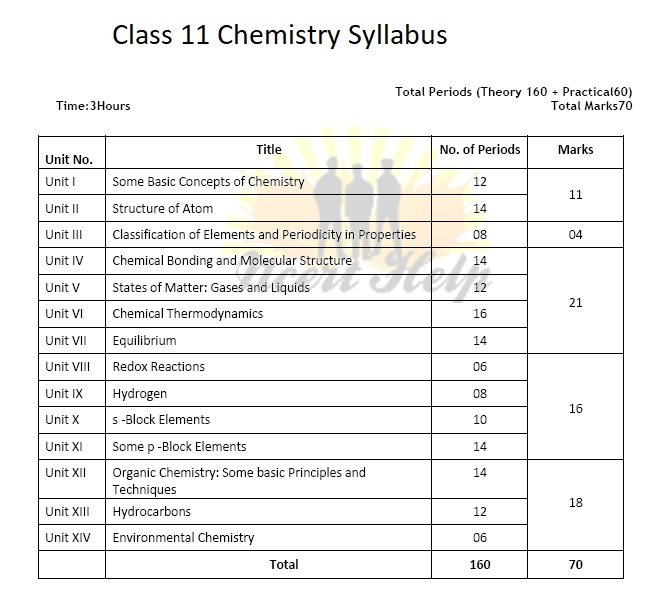
General Introduction: Importance and scope of Chemistry. Nature of matter, laws of chemical combination, Daltons atomic theory: concept of elements, atoms and molecules. Atomic and molecular masses, mole concept and molar mass, percentage composition, empirical and molecular formula, chemical reactions, stoichiometry and calculations based on stoichiometry.
Discovery of Electron, Proton and Neutron, atomic number, isotopes and isobars. Thomsons model and its limitations. Rutherfords model and its limitations, Bohrs model and its limitations, concept of shells and subshells, dual nature of matter and light, de Broglies relationship, Heisenberg uncertainty principle, concept of orbitals, quantum numbers, shapes of s, p and d orbitals, rules for filling electrons in orbitals - Aufbau principle, Paulis exclusion principle and Hunds rule, electronic configuration of atoms, stability of half-filled and completely filled orbitals.
Significance of classification, brief history of the development of periodic table, modern periodic law and the present form of periodic table, periodic trends in properties of elements -atomic radii, ionic radii, inert gas radii, Ionization enthalpy, electron gain enthalpy, electronegativity, valency. Nomenclature of elements with atomic number greater than 100.
Valence electrons, ionic bond, covalent bond, bond parameters, Lewis structure, polar character of covalent bond, covalent character of ionic bond, valence bond theory, resonance, geometry of covalent molecules, VSEPR theory, concept of hybridization, involving s, p and d orbitals and shapes of some simple molecules, molecular orbital theory of homonuclear diatomic molecules(qualitative idea only), Hydrogen bond.
Three states of matter, intermolecular interactions, types of bonding, melting and boiling points, role of gas laws in elucidating the concept of the molecule, Boyles law, Charles law, Gay Lussacs law, Avogadros law, ideal behaviour, empirical derivation of gas equation, Avogadros number, ideal gas equation. Deviation from ideal behaviour, liquefaction of gases, critical temperature, kinetic energy and molecular speeds (elementary idea), Liquid State- vapour pressure, viscosity and surface tension (qualitative idea only, no mathematical derivations)
Concepts of System and types of systems, surroundings, work, heat, energy, extensive and intensive properties, state functions. First law of thermodynamics -internal energy and enthalpy, heat capacity and specific heat, measurement of U and H, Hesss law of constant heat summation, enthalpy of bond dissociation, combustion, formation, atomization, sublimation, phase transition, ionization, solution and dilution. Second law of Thermodynamics (brief introduction) Introduction of entropy as a state function, Gibbs energy change for spontaneous and non- spontaneous processes, criteria for equilibrium. Third law of thermodynamics (brief introduction).
Equilibrium in physical and chemical processes, dynamic nature of equilibrium, law of mass action, equilibrium constant, factors affecting equilibrium - Le Chateliers principle, ionic equilibrium- ionization of acids and bases, strong and weak electrolytes, degree of ionization, ionization of poly basic acids, acid strength, concept of pH, hydrolysis of salts (elementary idea), buffer solution, Henderson Equation, solubility product, common ion effect (with illustrative examples).
Concept of oxidation and reduction, redox reactions, oxidation number, balancing redox reactions, in terms of loss and gain of electrons and change in oxidation number, applications of redox reactions.
Position of hydrogen in periodic table, occurrence, isotopes, preparation, properties and uses of hydrogen, hydrides-ionic covalent and interstitial; physical and chemical properties of water,heavy water, hydrogen peroxide -preparation, reactions and structure and use; hydrogen as a fuel.
Group 1 and Group 2 Elements General introduction, electronic configuration, occurrence, anomalous properties of the first element of each group, diagonal relationship, trends in the variation of properties (such as ionization enthalpy, atomic and ionic radii), trends in chemical reactivity with oxygen, water, hydrogen and halogens, uses. Preparation and Properties of Some Important Compounds: Sodium Carbonate, Sodium Chloride, Sodium Hydroxide and Sodium Hydrogen carbonate, Biological importance of Sodium and Potassium. Calcium Oxide and Calcium Carbonate and their industrial uses, biological importance of Magnesium and Calcium.
General Introduction to p -Block Elements Group 13 Elements: General introduction, electronic configuration, occurrence, variation of properties, oxidation states, trends in chemical reactivity, anomalous properties of first element of the group, Boron - physical and chemical properties, some important compounds: Borax, Boric acid, Boron Hydrides, Aluminium: Reactions with acids and alkalies, uses. Group 14 Elements: General introduction, electronic configuration, occurrence, variation of properties, oxidation states, trends in chemical reactivity, anomalous behaviour of first elements. Carbon-catenation, allotropic forms, physical and chemical properties; uses of some important compounds: oxides. Important compounds of Silicon and a few uses: Silicon Tetrachloride, Silicones, Silicates and Zeolites, their uses.
General introduction, methods of purification, qualitative and quantitative analysis, classification and IUPAC nomenclature of organic compounds. Electronic displacements in a covalent bond: inductive effect, electromeric effect, resonance and hyper conjugation. Homolytic and heterolytic fission of a covalent bond: free radicals, carbocations, carbanions, electrophiles and nucleophiles, types of organic reactions.
Classification of Hydrocarbons Aliphatic Hydrocarbons: Alkanes - Nomenclature, isomerism, conformation (ethane only), physical properties, chemical reactions including free radical mechanism of halogenation, combustion and pyrolysis. Alkenes - Nomenclature, structure of double bond (ethene), geometrical isomerism, physical properties, methods of preparation, chemical reactions: addition of hydrogen, halogen, water, hydrogen halides (Markovnikovs addition and peroxide effect), ozonolysis, oxidation, mechanism of electrophilic addition. Alkynes - Nomenclature, structure of triple bond (ethyne), physical properties, methods of preparation, chemical reactions: acidic character of alkynes, addition reaction of - hydrogen, halogens, hydrogen halides and water. Aromatic Hydrocarbons: Introduction, IUPAC nomenclature, benzene: resonance, aromaticity, chemical properties: mechanism of electrophilic substitution. Nitration, sulphonation, halogenation, Friedel Crafts alkylation and acylation, directive influence of functional group in monosubstituted benzene. Carcinogenicity and toxicity.
Environmental pollution - air, water and soil pollution, chemical reactions in atmosphere, smog, major atmospheric pollutants, acid rain, ozone and its reactions, effects of depletion of ozone layer, greenhouse effect and global warming- pollution due to industrial wastes, green chemistry as an alternative tool for reducing pollution, strategies for control of environmental pollution.
Class 11 chemistry syllabus, 11th chemistry syllabus, cbse class 11 chemistry syllabus, up board syllabus of class 11th chemistry pdf, chemistry syllabus for class 11 state board, changes in ncert chemistry class 11, ncert syllabus for class 11 chemistry, organic chemistry class 11 syllabus, cbse 11th chemistry syllabus, class 11 chemistry practical syllabus, up board class 11 chemistry syllabus, cbse class 11 science syllabus, isc class 11 chemistry syllabus, rbse syllabus for class 11
Copyright @ ncerthelp.com A free educational website for CBSE, ICSE and UP board.