
The present syllabus has been designed around seven broad themes viz. Food; Materials; The World of The Living; How Things Work; Moving Things, People and Ideas; Natural Phenomenon and Natural Resources. Special care has been taken to avoid temptation of adding too many concepts than can be comfortably learnt in the given time frame. No attempt has been made to be comprehensive. At this stage, while science is still a common subject, the disciplines of Physics, Chemistry and Biology begin to emerge. The students should be exposed to experiences based on hands on activities as well as modes of reasoning that are typical of the subject.
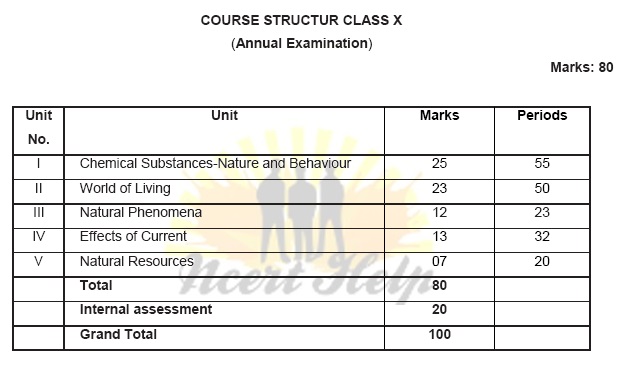
Unit I: Chemical Substances - Nature and Behaviour
Chemical reactions: Chemical equation, Balanced chemical equation, implications of a balanced chemical equation, types of chemical reactions: combination, decomposition, displacement, double displacement, precipitation, neutralization, oxidation and reduction.
Acids, bases and salts: Their definitions in terms of furnishing of H+ and OH– ions, General properties, examples and uses, concept of pH scale (Definition relating to logarithm not required), importance of pH in everyday life; preparation and uses of Sodium Hydroxide, Bleaching powder, Baking soda, Washing soda and Plaster of Paris.
Metals and nonmetals: Properties of metals and non-metals; Reactivity series; Formation and properties of ionic compounds; Basic metallurgical processes; Corrosion and its prevention.
Carbon compounds: Covalent bonding in carbon compounds. Versatile nature of carbon. Homologous series. Nomenclature of carbon compounds containing functional groups (halogens, alcohol, ketones, aldehydes, alkanes and alkynes), difference between saturated hydro carbons and unsaturated hydrocarbons. Chemical properties of carbon compounds (combustion, oxidation, addition and substitution reaction). Ethanol and Ethanoic acid (only properties and uses), soaps and detergents.
Periodic classification of elements: Need for classification, early attempts at classification of elements (Dobereiner’s Triads, Newland’s Law of Octaves, Mendeleev’s Periodic Table), Modern periodic table, gradation in properties, valency, atomic number, metallic and non-metallic properties.
Unit II: World of Living
Life processes: ‘Living Being’. Basic concept of nutrition, respiration, transport and excretion in plants and animals.
Control and co-ordination in animals and plants: Tropic movements in plants; Introduction of plant hormones; Control and co-ordination in animals: Nervous system; Voluntary, involuntary and reflex action; Chemical co-ordination: animal hormones.
Reproduction: Reproduction in animals and plants (asexual and sexual) reproductive health-need and methods of family planning. Safe sex vs HIV/AIDS. Child bearing and women’s health.
Heredity and Evolution: Heredity; Mendel’s contribution- Laws for inheritance of traits: Sex determination: brief introduction; Basic concepts of evolution.
Unit III: Natural Phenomena
Reflection of light by curved surfaces; Images formed by spherical mirrors, centre of curvature, principal axis, principal focus, focal length, mirror formula (Derivation not required), magnification.
Refraction; Laws of refraction, refractive index.
Refraction of light by spherical lens; Image formed by spherical lenses; Lens formula (Derivation not required); Magnification. Power of a lens.
Functioning of a lens in human eye, defects of vision and their corrections, applications of spherical mirrors and lenses.
Refraction of light through a prism, dispersion of light, scattering of light, applications in daily life.
Unit IV: Effects of Current
Electric current, potential difference and electric current. Ohm’s law; Resistance, Resistivity, Factors on which the resistance of a conductor depends. Series combination of resistors, parallel combination of resistors and its applications in daily life. Heating effect of electric current and its applications in daily life. Electric power, Interrelation between P, V, I and R.
Magnetic effects of current : Magnetic field, field lines, field due to a current carrying conductor, field due to current carrying coil or solenoid; Force on current carrying conductor, Fleming’s Left Hand Rule, Electric Motor, Electromagnetic induction. Induced potential difference, Induced current. Fleming’s Right Hand Rule, Electric Generator, Direct current. Alternating current: frequency of AC. Advantage of AC over DC. Domestic electric circuits.
Unit V: Natural Resources
Sources of energy: Different forms of energy, conventional and non-conventional sources of energy: Fossil fuels, solar energy; biogas; wind, water and tidal energy; Nuclear energy. Renewable versus non-renewable sources of Energy.
Our environment: Eco-system, Environmental problems, Ozone depletion, waste production and their solutions. Biodegradable and non-biodegradable substances.
Management of natural resources: Conservation and judicious use of natural resources. Forest and wild life; Coal and Petroleum conservation. Examples of people’s participation for conservation of natural resources. Big dams: advantages and limitations; alternatives, if any. Water harvesting. Sustainability of natural resources.
1. A. Finding the pH of the following samples by using pH paper/universal indicator: Unit-I
(i) Dilute Hydrochloric Acid
(ii) Dilute NaOH solution
(iii) Dilute Ethanoic Acid solution
(iv) Lemon juice
(v) Water
(vi) Dilute Hydrogen Carbonate solution
B. Studying the properties of acids and bases (HCl & NaOH) on the basis of their reaction with: Unit-I
a) Litmus solution (Blue/Red)
b) Zinc metal
c) Solid sodium carbonate
2. Performing and observing the following reactions and classifying them into: Unit-I
A. Combination reaction
B. Decomposition reaction
C. Displacement reaction
D. Double displacement reaction
(i) Action of water on quicklime
(ii) Action of heat on ferrous sulphate crystals
(iii) Iron nails kept in copper sulphate solution
(iv) Reaction between sodium sulphate and barium chloride solutions
3. Observing the action of Zn, Fe, Cu and Al metals on the following salt solutions: Unit-I
i) ZnSO4(aq)
ii) FeSO4(aq)
iii) CuSO4(aq)
iv) Al2 (SO4)3(aq)Arranging Zn, Fe, Cu and Al (metals) in the decreasing order of reactivity based on the above result.
4. Studying the dependence of potential difference (V) across a resistor on the current (I) passing through it and determine its resistance. Also plotting a graph between V and I. Unit-IV
5. Determination of the equivalent resistance of two resistors when connected in series and parallel. Unit-IV
6. Preparing a temporary mount of a leaf peel to show stomata. Unit- II
7. Experimentally show that carbon dioxide is given out during respiration. Unit-II
8. Study of the following properties of acetic acid (ethanoic acid): Unit- I
i) Odour
ii) solubility in water
iii) effect on litmus
iv) reaction with Sodium Hydrogen Carbonate
9. Study of the comparative cleaning capacity of a sample of soap in soft and hard water. Unit- I
10. Determination of the focal length of: Unit-III
i) Concave mirror
ii) Convex lensby obtaining the image of a distant object.
11. Tracing the path of a ray of light passing through a rectangular glass slab for different angles of incidence. Measure the angle of incidence, angle of refraction, angle of emergence and interpret the result. Unit - III
12. Studying (a) binary fission in Amoeba, and (b) budding in yeast and Hydra with the help of prepared slides. Unit-II
13. Tracing the path of the rays of light through a glass prism. Unit-III
14. Finding the image distance for varying object distances in case of a convex lens and drawing corresponding ray diagrams to show the nature of image formed. Unit-III
15. Identification of the different parts of an embryo of a dicot seed (Pea, gram or red kidney bean).Unit-II
Syllabus class 10 science, cbse syllabus for class 10 science, social science class 10 syllabus, cbse 10th science syllabus, ncert syllabus for class 10 science, up board class 10 science syllabus, 10th science syllabus, class 10 science practical syllabus, 10th new syllabus science, 10th physics syllabus, cbse 10th physics syllabus, icse class 10 chemistry syllabus, marks distribution of science class 10, cbse class 10 biology syllabus, cbse class 10 chemistry syllabus
Copyright @ ncerthelp.com A free educational website for CBSE, ICSE and UP board.